Katharina M. Fromm
University of Fribourg, Switzerland
Title: Silver compounds and nanorattles: triggered drug delivery and prevention of implant infections
Biography
Biography: Katharina M. Fromm
Abstract
Statement of the Problem: More and more artificial materials are implanted into the human body to replace bone and organ function in case of failure. These materials do not possess a natural defence system and are hence prone to bacterial adhesion which can occur during operation or via hematogenous seeding. Bacterial infections of implants may have severe consequences as they cannot be treated with traditional antibiotics.
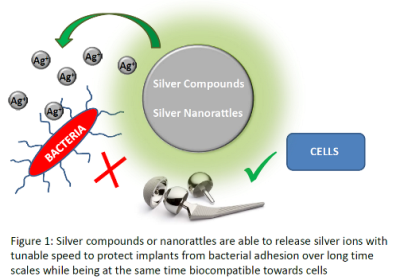
Methodology & Theoretical Orientation: We developed coordination compounds based on ionic silver and a ligand to be attached on metallic implant surfaces. In parallel, silver nanoparticle containing nanorattles based on inorganic shell materials such as silica or titania were developed for a slow silver ion release. The compounds were thoroughly characterized in bulk as well as in the form of surface coatings. The antimicrobial activities were evaluated by in vitro Kirby Bauer tests as well as in vivo tests. Biocompatibility tests were performed on fibroblast cells in vivo. Both results were related to the silver ion release profiles from the materials in biological buffer media. A bacterial sensor has been developed to link the drug release from nanorattles to the presence of bacteria.
Findings: We have found several new coordination compounds based on silver ions that can be successfully attached to metallic implant surfaces to release antimicrobial metal ions while remaining biocompatible. For a prolonged release, we developed nanocontainers that provide drug protection and tunable release times and that can also be attached to implant surfaces. In order to target the release in a more controlled way, we developed a bacterial sensor able to recognize DNA material in a selective, fast and sensitive way for a specific treatment.
Conclusion & Significance: With the panoply of antimicrobial compounds, implant materials can be protected immediately after operation to prevent infections occurring during this process, as well as long-term protected against infections occurring via hematogenous seeding. The bacterial sensor allows to trigger drug release and can be used as analytical tool as well for rapid and specific diagnostics